Applications for Medical Device Investigational Testing Authorizations Guidance Document
$ 16.50 · 4.8 (138) · In stock

Applications for Medical Device Investigational Testing Authorizations Guidance Document
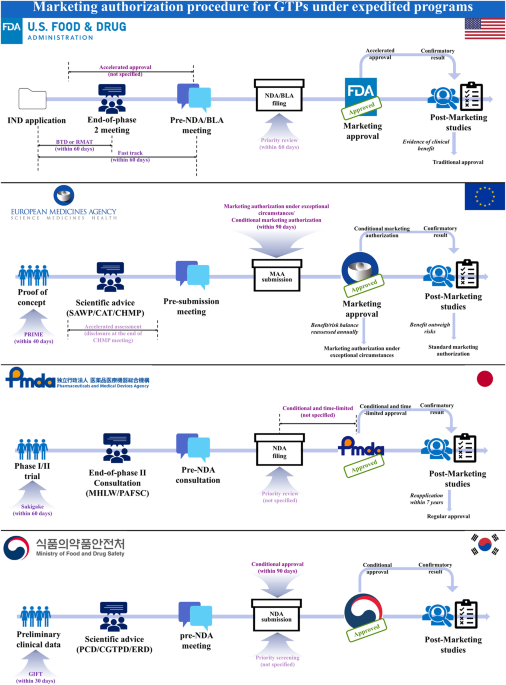
Analysis and comparative evaluation of expedited programs for gene therapy products: insights from the United States, the European Union, Japan, and South Korea
List of documents to upload to the Spanish Agency of Medicines and

Regulatory oversight of genetic testing in Canada: Health Canada perspective
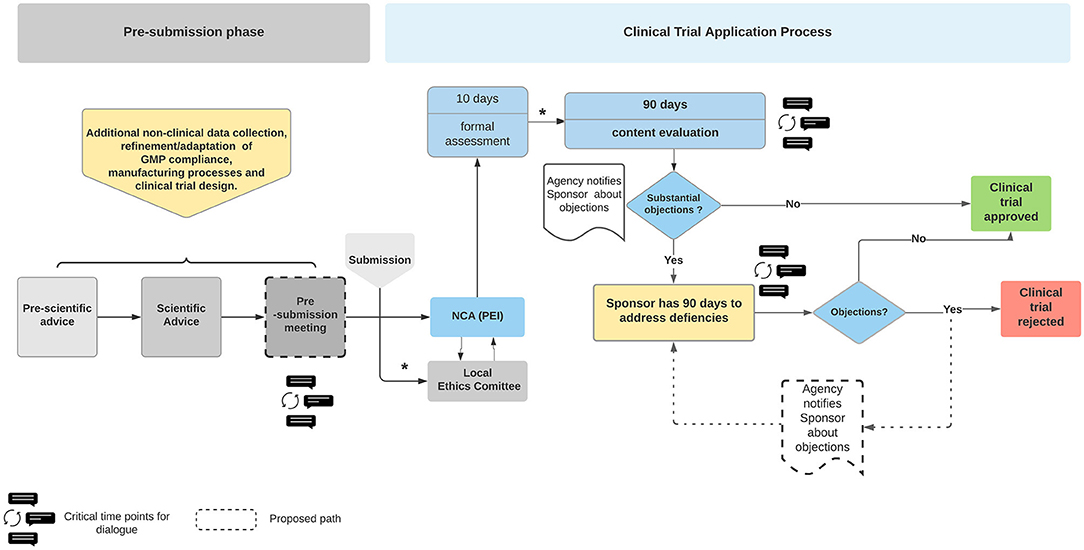
Frontiers Advanced Therapy Medicinal Products' Translation in Europe: A Developers' Perspective

Applications for Medical Device Investigational Testing Authorizations Guidance Document

FDA Guidance on Dual 510(k) and CLIA Waivers

Frontiers A Regulatory Risk-Based Approach to ATMP/CGT Development: Integrating Scientific Challenges With Current Regulatory Expectations

Current practices and reform proposals for the regulation of
-image.jpg)
When does My Application Qualify for an Abbreviated 510(k)?
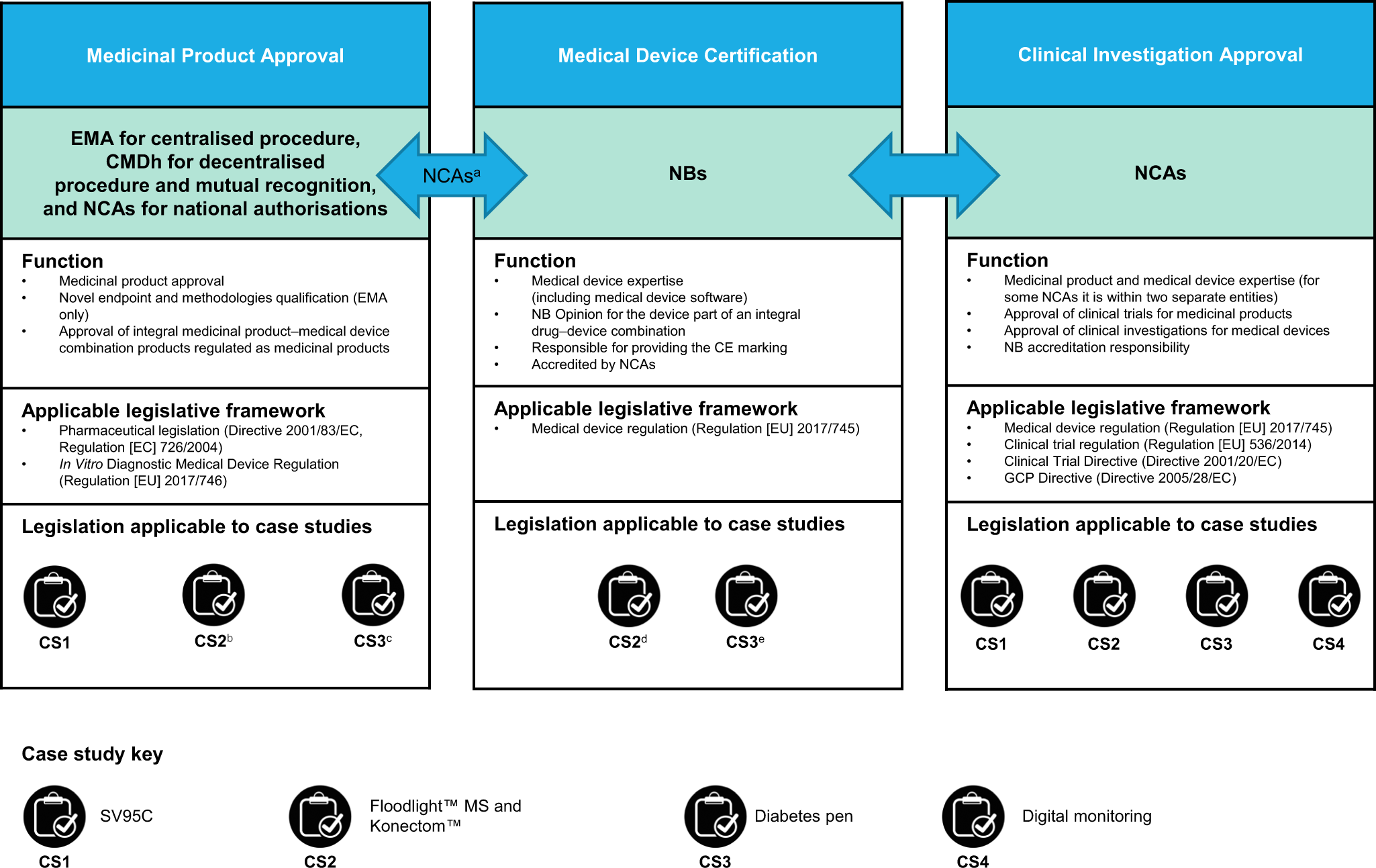
Evolving regulatory perspectives on digital health technologies

Pathways to a FDA Approved or Cleared Medical Device - StarFish Medical

Regulatory oversight of genetic testing in Canada: Health Canada